Get the Expert Guidance You Need Across the Board.
With over a century of combined experience, Sterling IRB’s board of professionals from the medical, scientific, and legal industries are committed to delivering high-quality, ethical reviews that move your research toward regulatory approval.
The decisions of the board are guided by the ethical principles set forth in the Belmont Report. The board reviews clinical research protocols, participant informed consent documents, and investigator credentials to ensure compliance with federal regulations. It also ensures that research subjects are appropriately informed about the risks and benefits of participating in a research study. Upon receiving initial approval from the board, investigators and sites are monitored through the continuing review process of all study-related materials including, but not limited to, the review of ongoing reports, monitoring letters, safety information documents, protocol amendments, informed consent document revisions, and recruitment/retention materials.
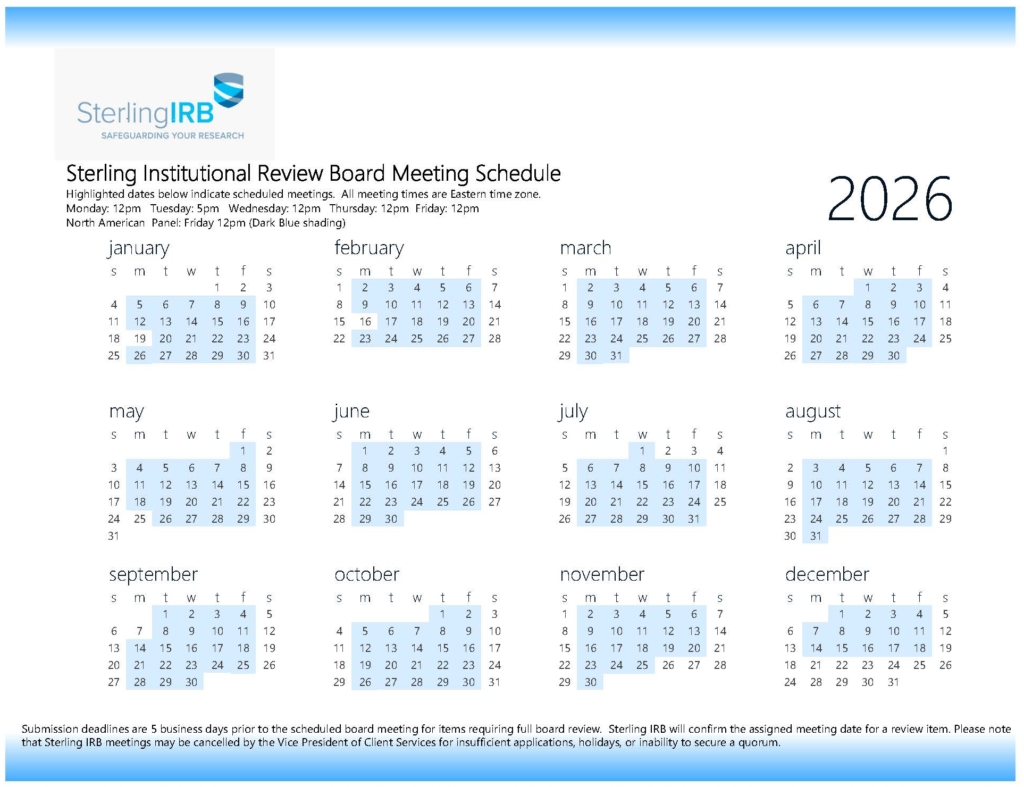
Sterling IRB convenes daily, Monday through Friday, and requests that items requiring full board IRB review be submitted within four to five business days of a board meeting. Upon receipt of a submission, Sterling IRB will confirm the assigned meeting date for a review item. Please let us know your timelines; we will do our very best to accommodate your requirements.
Board Roster
The Sterling Institutional Review Board consists of a diverse group of scientific and nonscientific members with nearly a century of collective IRB experience. Meeting all requirements of the federal regulations for IRB membership, our board candidates complete a formal mentoring program before consideration for membership. Board members are provided with ongoing education and are required to complete monthly trainings in human subject protection.
Accreditation
AAHRPP Accredited
Certified IRB Professionals
SMART IRB Participating Institution